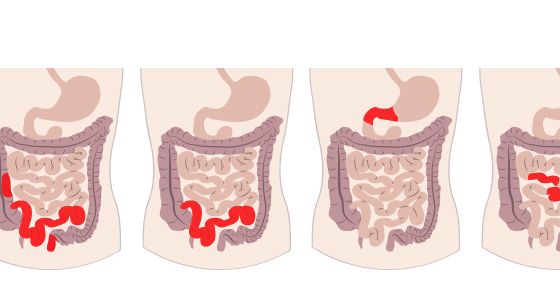
In simple terms faecal microbiota transplantation, or FMT as it is often referred to, is the process of taking faeces from a healthy donor, extracting the beneficial intestinal bacteria and yeasts from it and then transplanting it into the colon of a recipient. It is also sometimes referred to as a stool transplant.
In recent years much research has pointed to the importance of a diverse microbiota to maintain human health. In FMT the recipient’s current microbiota is populated with the bacteria extracted from the donor, thus giving them a new gut microbiome make-up.
The use of FMT has been cited in Chinese literature for over 2000 years and since the 1950s in western literature. However, the use of FMT to treat medical conditions is an emerging area of research and medicine and there is currently limited data on outcomes and safety. Studies have shown that FMT is a useful treatment for people with Clostridium difficile (C.diff). In one study FMT was successful in resolving recurrent C.diff infections after one infusion in 81% of patients1. As a result FMT is now an approved treatment for for recurrent C.diff infections in the UK.
FMT has also been used in people with a range of different gut conditions such as Crohn’s disease, ulcerative colitis, irritable bowel syndrome (IBS) and chronic diarrhoea and/or constipation. It has also been used in people who have neurological conditions, such chronic fatigue syndrome (CFS), multiple sclerosis (MS) and Parkinson’s Disease. However, evidence for its effectiveness in these areas is still emerging.
Since the late 80s FMT trials have been carried out on patients with ulcerative colitis. In 1989 55 patients with various gastrointestinal disorders were given faecal microbiota transplants and a follow up of six patients in 2003 who had UC showed there was ‘no clinical, colonoscopic or histologic evidence of UC in any patient’2.
A review published in August 20183 of the FMT trials to date said: ‘Clinical trials data are still poor but strongly support a future introduction of FMT in therapy to manage IBD microbiome. More studies are needed to assess the optimal route of administration and the frequency of FMT, the best matched donor for each patient as well as the risks associated with FMT in IBD’.
Much of the evidence for FMT use to treat inflammatory bowel disease (IBD) appears to be for ulcerative colitis, rather than Crohn’s disease.
In an interview in Gastroenterology & Hepatology in May 2018 Dr Paul Moayyedi said: “A small amount of observational case-series data, as well as several randomized, controlled trials (including one conducted by my colleagues and I), suggest that FMT may work in the setting of ulcerative colitis. There is less evidence that FMT works in patients with Crohn’s disease, but the same principle applies”.4
A review in 2017 of four of these trials on patients with UC5 concluded: ‘Among randomized controlled trials, short-term use of FMT shows promise as a treatment to induce remission in active UC based on the efficacy and safety observed. However, there remain many unanswered questions that require further research before FMT can be considered for use in clinical practice’.
A study examining whether certain types of bacteria were more likely to make FMT an effective treatment for UC. The study found that patients in remission after FMT had enrichment of Eubacterium hallii and Roseburia inulivorans bacteria and increased levels of short-chain fatty acid biosynthesis and secondary bile acids6.
At the moment FMT for IBD isn't widely available.
Researchers have tried various different methods to carry out transplants. These include:
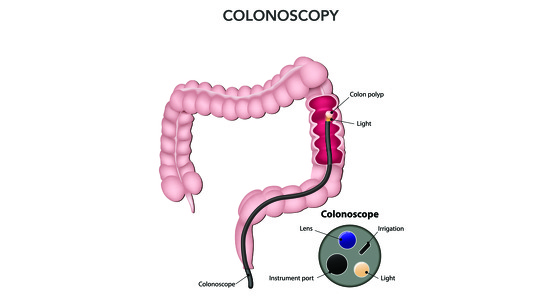
Faecal microbiota transplants for C.diff tend to be carried out via colonoscopy
FMT donors are rigorously screened to make sure they are suitable. The donor must be healthy and lead a healthy lifestyle. There are stool banks which a donor may be selected from.
The full extent of risks when using FMT for treating IBD aren’t fully known yet.
When FMT is used to treat C.diff one of the main associated risks is perforation of the bowel if colonoscopy is used to deliver the faecal preparation to the bowel.
1. van Nood E, Vrieze A, Nieuwdorp M, Fuentes S, Zoetendal EG, de Vos WM, Visser CE, Kuijper EJ, Bartelsman JF, Tijssen JG, Speelman P, Dijkgraaf MG, Keller JJ (January 2013). "Duodenal infusion of donor feces for recurrent Clostridium difficile". The New England Journal of Medicine. 368 (5): 407‐15.
2. Borody TJ, Warren EF, Leis S, Surace R, Ashman O. Treatment of ulcerative colitis using fecal bacteriotherapy. J Clin Gastroenterol. 2003 Jul;37(1):42-7.
3. Tagore Sunkara, Prashanth Rawla, Andrew Ofosu, Vinaya Gaduputi. Fecal microbiota transplant ‐ a new frontier in inflammatory bowel disease. Journal of Inflammation Research. 31 August 2018 Volume 2018:11 Pages 321””328
4. Paul Moayyedi. Update on Fecal Microbiota Transplantation in Patients With Inflammatory Bowel Disease. Gastroenterology & Hepatology. May 2018 - Volume 14, Issue 5
5. Narula N, Kassam Z, Yuan Y, Colombel JF, Ponsioen C, Reinisch W, Moayyedi P. Systematic Review and Meta-analysis: Fecal Microbiota Transplantation for Treatment of Active Ulcerative Colitis. Inflamm Bowel Dis. 2017 Oct;23(10):1702-1709.
6. Sudarshan Paramsothy et al. Specific Bacteria and Metabolites Associated with Response to Fecal Microbiota Transplantation in Patients with Ulcerative Colitis. Gastroenterology, December 2018. https://doi.org/10.1053/j.gastro.2018.12.001