
In this article we take a look at Symprove and IBD.
Read about the reasons why you may want to introduce probiotic bacteria to your body and what your microbiome is.
Symprove* is a water-based drink that contains four strains of live, activated bacteria -. Each 60ml shot contains around 10 billion of these bacteria:
It also contains:
It is gluten free and dairy free and suitable for vegans, although it is manufactured using barley extract (which has gluten in it) which is then removed from the product.
It comes in several flavours, including original, mango and passionfruit, and strawberry and raspberry (the flavoured versions contains artificial sweeteners).
Symprove is a liquid drink taken on an empty stomach 10 minutes before eating
Symprove say it works because:
Some research has been done which supports these claims. A study comparing eight commercially available probiotics (Actimel, Align, Symprove, Biobalance, Probio7, Yakult, VSL#3 and Bio-Kult)1. In the study (published in March 2015) the eight probiotics were put through three viability tests - to see if the bacteria was in a live state before being taken, if the bacteria survived the transit to the stomach and if the bacteria then thrived in the targeted area of the gut. Symprove was the only probiotic of the eight to pass all three tests. These tests were carried out 'in vitro’ (test tube experiments).
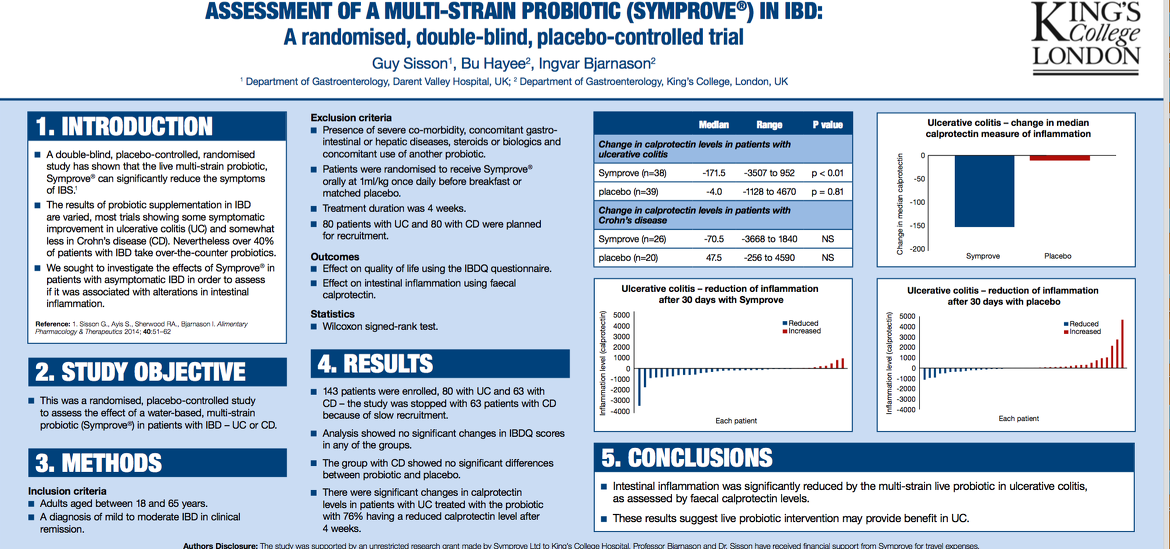
A recent research study was carried out into the use of Symprove and ulcerative colitis (UC)2. It found that 76% of those with ulcerative colitis taking part in the study had reductions in faecal levels of calprotectin - elevated levels of calprotectin indicate intestinal inflammation.
There are no studies in relation to Symprove and Crohn's disease or other forms of IBD.
A study into Symprove and irritable bowel syndrome (IBS) was published in 20143. The study was completed by 152 people symptomatic with IBS who took either Symprove or a placebo for 12 weeks. At the end of the 12 weeks a statistically significant improvement in overall symptom severity of symptoms in those who took Symprove was seen.
No other studies have been carried out into Symprove and other conditions.
Symprove is made by Symprove Holdings Ltd. Symprove was founded by Barry Smith, a UK (Surrey) farmer who created Symprove to help his farm animals. It was so successful he decided to see if it worked for humans too. The human trials were successful and Symprove started being manufactured for human consumption.
It is all made on site at Symprove’s factory. Symprove sources barley from local growers in Surrey and Hampshire. This barley grain is then germinated. The germinated barley is rolled and water is added. It goes through a series of temperature changes. Food structures (such as amino acids, vitamins and enzymes) are extracted which is then pasturised and the four strains of bacteria used by Symprove are added. Symprove has the bacteria grown for them from the core stock used originally to ensure purity of the strain. After 24 hours the bacteria has reached optimal numbers and it is ready to be bottled. The whole process - from germination to bottling takes 10 days.
Symprove says that during the production process any gluten in the barley is removed - through germination, pasteurisation and filtration. Samples are also sent to be checked to ensure Symprove remains under the 20 parts per million limits for a gluten free product.
Samples are sent to an external GMP (Good Manufacturing Practices) accredited laboratory to verify the exact strains of bacteria grown and check there are no other bacteria or contaminants in the product. To pass the tests there must also be in the region of 10 billion live activated colony forming bacteria units per 50ml serving.

Symprove suggests you take a full dose (60ml) daily for 12 weeks. After this time they believe you should see results. After 12 weeks you can choose whether to take a maintenance dose’ (of anywhere up to 60ml) each day.
Unopened bottles should be stored in a cool, dark place. Once opened they should be refrigerated. It has a shelf-life of four months.
Symprove is made in the UK and is considered to be a food supplement (it is not a medicine).
In the UK most probiotics are governed under the same laws as foods, not medicines, and governing bodies include the European Food Safety Authority (EFSA), the Food Safety Authority (FSA) and the Advisory Committee on Borderline Substances.
These bodies issue guidelines around what probiotic companies can say in relation to their product and health claims they can make. Even if a probiotic has medical evidence to support its use for a certain condition they cannot publicise this unless they go through strict medical testing - a very expensive and lengthy process.
This is why you will often find that probiotic companies do not mention specific health conditions on their website and marketing material. Guidelines have also been issued to say that the term probiotic may no longer be used in the promotion of supplements by the companies. The Advertising Standards Authority also has oversight regarding claims made and any complaints arising. In general the industry is self-regulated so some companies choose to ‘bend’ the rules around making health claims in relation to their product.
A one month supply of Symprove costs £79 or 12 weeks is £149.99. They also offer monthly subscriptions for £39.99 (when you sign up to 12 months) or £49.99 for a monthly rolling subscription that you can cancel at any time. Symprove is available for shipping outside of the UK.
If you enter code IBDRELIEF50 at checkout you'll get 50% off the first three months of a monthly rolling subscription.
You can buy it directly from the Symprove website
More information is available at www.symprove.com*
*Please note this is an affiliate link which means if you buy from the company we may receive a commission. You will pay the same price and any commission gained will be used to support IBDrelief's work.